Are you reprocessing your instrument containers correctly? – Part 4 Copy
Quality inspections
Inspecting rigid containers after cleaning and disinfection is as important as the cleaning itself. Inspection checks for residual soils and cleaning chemistry, wear and tear, and mechanical disfunction that could potentially interfere with sterilization or could fail to maintain sterility of the contents. If inspection identifies damaged containers or accessories, they need to be repaired or replaced. Items to inspect include (but are not limited to) gaskets, valves, interior baskets, mating surfaces, reusable filters, filter retainers and latches. Damaged containers and accessories must be removed from the use inventory and the manufacturer needs to be consulted.
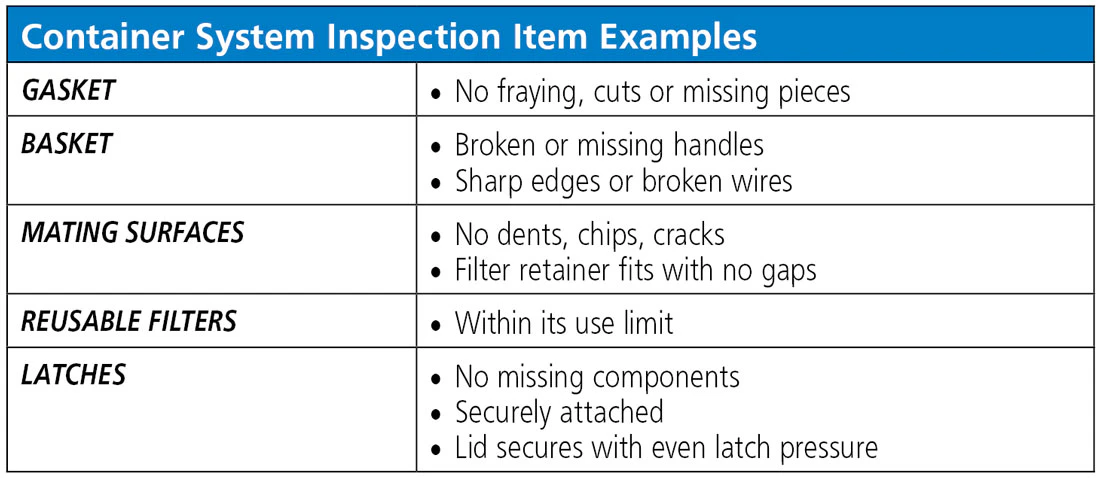
As part of a thorough quality system for containers, a routine preventive maintenance schedule should be established and documented; this assures that tools are in place to detect and/or avoid potential problems. Containers are not meant to last forever, so the container vendor can provide information about the expected useful life of each type of container. The department or purchasing manager should then budget accordingly for timely replacement of compromised or aging rigid containers.
Space constraints
Regulatory inspectors will not allow space constraints as an excuse for skipping reprocessing steps or not fully following manufacturers’ IFU. They will expect departments to maximize the space they have by performing proper planning that includes workflow, case demands and calculated expected throughputs. It’s worthwhile to consider automating the container cleaning process. Automation allows consistent proper practice while freeing staff to focus on case sets and other instrumentation.
Tight reprocessing spaces, like those in ambulatory surgical centers, require equipment with small footprints. In addition to the washer, it’s important to have the correct number and types of racks needed to process the containers in use at the facility. Depending on the case load, it may also be wise to invest in a small cart washer, which would allow the washer disinfector to be dedicated to instrumentation and the larger cart washer to case carts. This would enable a better use of space and equipment, and would support a continuous, more efficient workflow and throughput for both instruments and container systems.
Avoid container woes
No one wants to be the person standing in front of the auditor explaining why containers are not being properly processed. Rigid sterilization container systems are Class II medical devices and are cleared by the U.S. Food and Drug Administration (FDA) for use. Per FDA and other regulatory guidance, facilities must adhere to the container manufacturers’ IFU, which provide all the necessary information for correct use, care, cleaning, inspections, routine maintenance, sterilization methods, and storage. Facilities must develop policies and a standardized workflow that support these instructions, and confirm staff competency through training, education and regular evaluation of staff performance. Be the one who finds and corrects any problems in container processing, before the auditor does.
Download article & test
References
1. ANSI/AAMI ST41: 2008/(R)2012 Ethylene oxide sterilization in health care facilities: safety and effectiveness. Arlington, VA: Association for the Advancement of Medical Instrumentation
2. ANSI/AAMI ST58: 2013 Chemical sterilization and high-level disinfection in healthcare facilities. Arlington, VA: Association for the Advancement of Medical Instrumentation
3. ANSI/AAMI ST77: 2013. Containment devices for reusable medical device sterilization. Arlington, VA: Association for the Advancement of Medical Instrumentation.
4. ANSI/AAMI ST79: 2017. Comprehensive guide to steam sterilization and sterility assurance in healthcare facilities. Arlington, VA: Association for the Advancement of Medical Instrumentation.
5. Association of periOperative Registered Nurses: 2020. Guidelines for Perioperative Practice; Packaging Systems.
6. Shaffer HL, Harnish DA, McDonald M., et al. Sterility maintenance study: Dynamic evaluation of sterilized rigid containers and wrapped instrument trays to prevent bacterial ingress. American Journal Infection Control. 2015; 43; 1336-41
