Get the entire team trained! Click here to purchase group access.
If you need to purchase multiple courses for your group, click here.
CBSPD approved through May 31, 2025
HSPA approved through May 4, 2023
Learning Objectives:
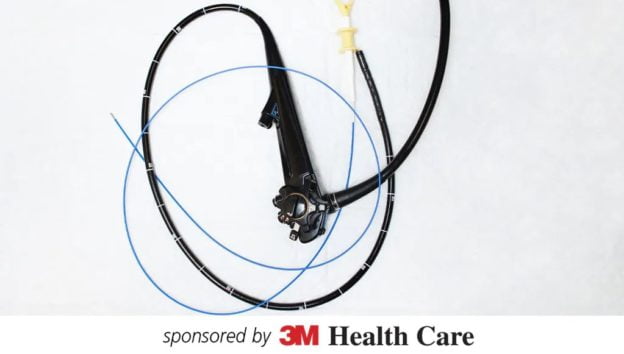
- Explain the differences of the sterility assurance levels for HLD as opposed to sterilization.
- Discuss recommendations for the disinfection and sterilization of flexible endoscopes.
- Explain the difference in storage time between high-level disinfected and sterilized scopes.
- Discuss steps to take when transitioning from HLD to sterilization of flexible endoscopes.
About the Instructor:

Login
Accessing this course requires a login. Please enter your credentials below!